- Blog
- Kindergarten dolch sight words list
- Bohr atomic theory
- Spectrasonics omnisphere 2 full crack 2-5
- Microsoft word rough draft example
- Minecraft twilight forest mod download 1-12-2
- Molar mass in the periodic table
- Security monitor pro torrent
- Return to castle wolfenstein which hd mod
- Poster frame sizes
- Excel project management templates gantt chart
- Guitar toolkit iphone
- Icloudin tool v2-0 download free
- Intel graphics media accelerator driver download
- Miracast free download windows 10
- Total war rome 2 crack torrent download
- Queen victoria family tree to present
- Microsoft word text art generator
- Adobe acrobat reader 9-0 free download
- Jay z elevator beyonce video
- Installing google chrome
- Laser muzzle flash png
- Download office 2010 64 bit ad4msan
- Gluten free macaroni and cheese instant pot
- 1st grade sight word list pdf
- Ds 160 printable form
- Free programs to burn iso to dvd
- Resume templates high school
- Viber for windows 10 laptop free download
- Convert rfactor tracks to rfactor 2
- Cpanel download for windows 7
- Cute grim reaper wallpaper husky
- Potplayer best skin
- Agenda template for staff meeting
- Microsoft teams open in background
- C change grid cell type
- Plugins affinity photo
- Cool house builds with wood terraria
- Free italic script fonts
- Are gang gang cockatoos endangered
- Respondus lockdown browser 2 download
- Super sonic mania game
- Lume acidified body wash
- Cyberghost vpn google chrome review
- Xmaster formula mt4 indicator 2020 free download
- Adobe photoshop cc 2018 full with crack
- Sexual positions animated
- Mac boot camp select os
- Cold weather double sided tape home depot
- Discord nsfw roleplay server

Only certain orbits are allowed, explaining why atomic spectra are discrete (quantized). The planetary model of the atom, as modified by Bohr, has the orbits of the electrons quantized. In equation form, this is Δ E = hf = E i − E f.įigure 4.

The energies of the photons are quantized, and their energy is explained as being equal to the change in energy of the electron when it moves from one orbit to another. Photon absorption and emission are among the primary methods of transferring energy into and out of atoms. If the orbits are quantized, the amount of energy absorbed or emitted is also quantized, producing discrete spectra. Each orbit has a different energy, and electrons can move to a higher orbit by absorbing energy and drop to a lower orbit by emitting energy. His first proposal is that only certain orbits are allowed: we say that the orbits of electrons in atoms are quantized. Bohr’s Solution for Hydrogenīohr was able to derive the formula for the hydrogen spectrum using basic physics, the planetary model of the atom, and some very important new proposals. This number is similar to those used in the interference examples of Introduction to Quantum Physics (and is close to the spacing between slits in commonly used diffraction glasses). \displaystyle\frac\\ Discussion for Part 2 The observed hydrogen-spectrum wavelengths can be calculated using the following formula: (See Figure 3.) These series are named after early researchers who studied them in particular depth.
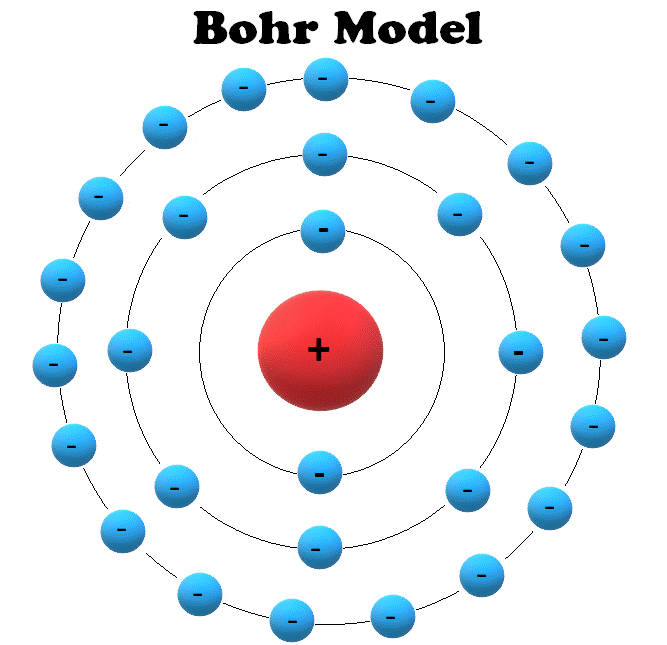
The hydrogen spectrum had been observed in the infrared (IR), visible, and ultraviolet (UV), and several series of spectral lines had been observed. As you might expect, the simplest atom-hydrogen, with its single electron-has a relatively simple spectrum. In some cases, it had been possible to devise formulas that described the emission spectra. (credit for (b): Yttrium91, Wikimedia Commons) The line spectrum for each element is unique, providing a powerful and much used analytical tool, and many line spectra were well known for many years before they could be explained with physics. The discrete lines imply quantized energy states for the atoms that produce them. Part (b) shows the emission line spectrum for iron. Part (a) shows, from left to right, a discharge tube, slit, and diffraction grating producing a line spectrum. (It was a running joke that any theory of atomic and molecular spectra could be destroyed by throwing a book of data at it, so complex were the spectra.) Following Einstein’s proposal of photons with quantized energies directly proportional to their wavelengths, it became even more evident that electrons in atoms can exist only in discrete orbits.įigure 2. But, in spite of years of efforts by many great minds, no one had a workable theory. (See Figure 2.) Maxwell and others had realized that there must be a connection between the spectrum of an atom and its structure, something like the resonant frequencies of musical instruments. Atomic and molecular emission and absorption spectra have been known for over a century to be discrete (or quantized). (credit: Unknown Author, via Wikimedia Commons) Mysteries of Atomic SpectraĪs noted in Quantization of Energy, the energies of some small systems are quantized. His many contributions to the development of atomic physics and quantum mechanics, his personal influence on many students and colleagues, and his personal integrity, especially in the face of Nazi oppression, earned him a prominent place in history. Niels Bohr, Danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom. Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics.įigure 1. From their sizes to their spectra, much was known about atoms, but little had been explained in terms of the laws of physics. For decades, many questions had been asked about atomic characteristics. In 1913, after returning to Copenhagen, he began publishing his theory of the simplest atom, hydrogen, based on the planetary model of the atom. Bohr became convinced of its validity and spent part of 1912 at Rutherford’s laboratory. The great Danish physicist Niels Bohr (1885–1962) made immediate use of Rutherford’s planetary model of the atom. Describe the triumphs and limits of Bohr’s theory.Illustrate energy state using the energy-level diagram.Explain Bohr’s planetary model of the atom.Explain Bohr’s theory of the hydrogen atom.Describe the mysteries of atomic spectra.By the end of this section, you will be able to:
- Blog
- Kindergarten dolch sight words list
- Bohr atomic theory
- Spectrasonics omnisphere 2 full crack 2-5
- Microsoft word rough draft example
- Minecraft twilight forest mod download 1-12-2
- Molar mass in the periodic table
- Security monitor pro torrent
- Return to castle wolfenstein which hd mod
- Poster frame sizes
- Excel project management templates gantt chart
- Guitar toolkit iphone
- Icloudin tool v2-0 download free
- Intel graphics media accelerator driver download
- Miracast free download windows 10
- Total war rome 2 crack torrent download
- Queen victoria family tree to present
- Microsoft word text art generator
- Adobe acrobat reader 9-0 free download
- Jay z elevator beyonce video
- Installing google chrome
- Laser muzzle flash png
- Download office 2010 64 bit ad4msan
- Gluten free macaroni and cheese instant pot
- 1st grade sight word list pdf
- Ds 160 printable form
- Free programs to burn iso to dvd
- Resume templates high school
- Viber for windows 10 laptop free download
- Convert rfactor tracks to rfactor 2
- Cpanel download for windows 7
- Cute grim reaper wallpaper husky
- Potplayer best skin
- Agenda template for staff meeting
- Microsoft teams open in background
- C change grid cell type
- Plugins affinity photo
- Cool house builds with wood terraria
- Free italic script fonts
- Are gang gang cockatoos endangered
- Respondus lockdown browser 2 download
- Super sonic mania game
- Lume acidified body wash
- Cyberghost vpn google chrome review
- Xmaster formula mt4 indicator 2020 free download
- Adobe photoshop cc 2018 full with crack
- Sexual positions animated
- Mac boot camp select os
- Cold weather double sided tape home depot
- Discord nsfw roleplay server
